Non-Surgical Hair Loss Treatment: An Evidence-Tiered 2026 Guide
Non-surgical hair loss treatment ranked by evidence: finasteride and minoxidil at the top, PRP in the middle, exosomes and stem cells largely unproven.
Medical disclaimer. This article is educational and not medical advice. Hair restoration outcomes are individual; only a qualified clinician can assess your case in a personal consultation, prescribe medication, and discuss side-effect profiles.
Quick answer
Non-surgical hair loss treatment in 2026 is best understood as a tiered hierarchy by evidence quality, not by marketing prominence. Tier 1 — finasteride and topical minoxidil — has Cochrane-level RCT support and produces measurable density gains in roughly 80–90% of treated men with androgenetic alopecia. Tier 1.5 — low-dose oral minoxidil — has a growing dermatology evidence base (Sinclair, Vañó-Galván) and is now standard practice in specialist centres. Tier 2 — PRP — has mixed RCT results and works as an adjunct, not a replacement. Tier 3 — LLLT, mesotherapy — has modest-to-weak evidence. Tier 4 — exosomes, stem cells, “hair vitamins” without deficiency — has marketing without comparable evidence. This pillar walks each tier honestly.
Table of contents
- The evidence-tiered framework
- Tier 1: finasteride and topical minoxidil (the foundation)
- Tier 1.5: low-dose oral minoxidil (the new standard)
- Tier 2: PRP (the modest tool)
- Tier 3: LLLT and mesotherapy (the variable adjuncts)
- Tier 4: exosomes, stem cells, and supplements without deficiency
- How to combine treatments (clinical protocols)
- When non-surgical isn’t enough
- Side-effect transparency
- How BergemHealth approaches non-surgical treatment
- What to do next
- Frequently asked questions
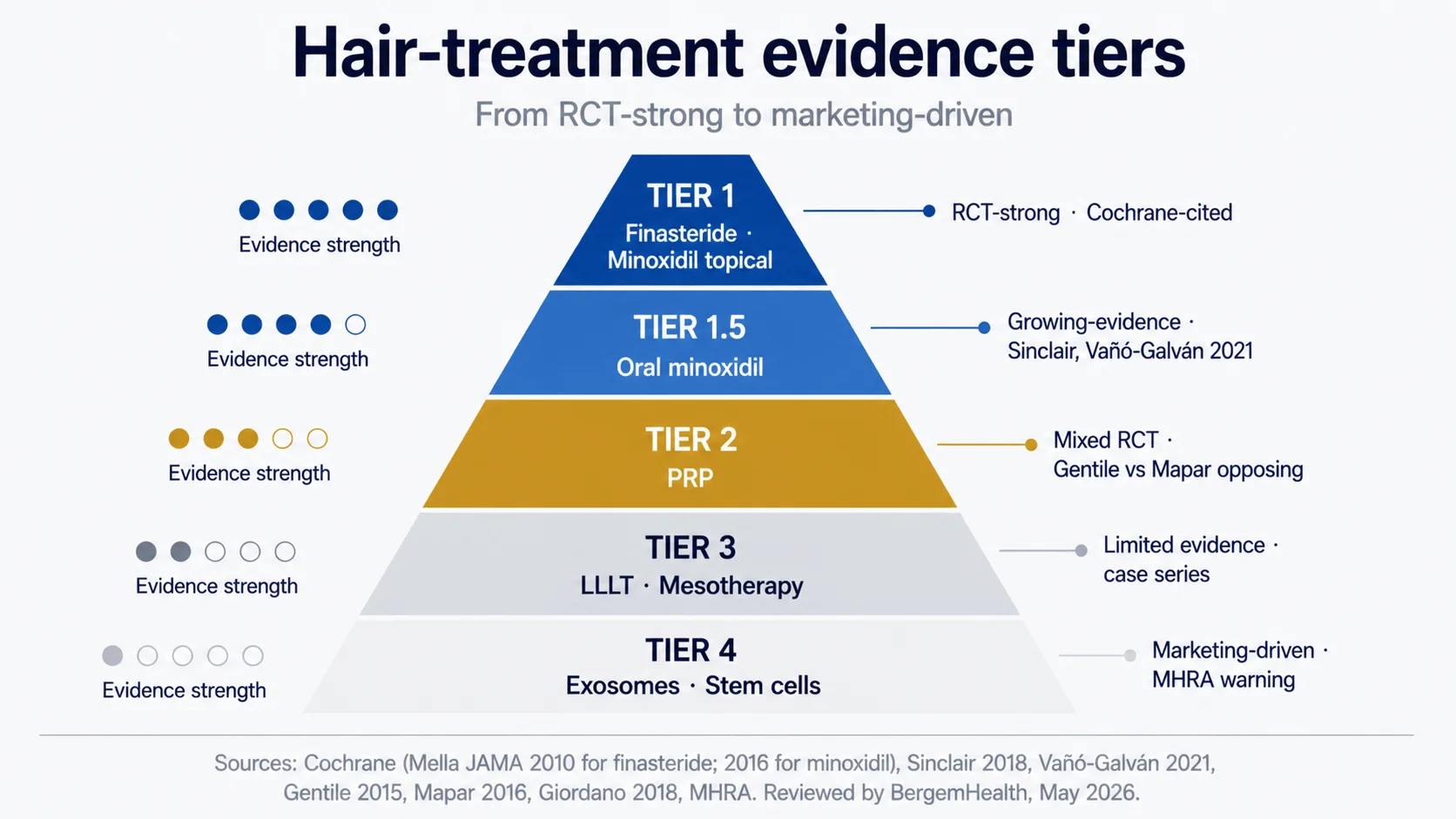
The evidence-tiered framework
Definition. Evidence-tiered treatment ranks interventions by the quality of clinical evidence supporting their efficacy. The top of the tier is reserved for therapies with multiple randomised controlled trials, ideally synthesised by Cochrane or equivalent. The bottom contains interventions with mostly mechanism-based reasoning, anecdotal results, or case series without controlled comparison. Marketing intensity and evidence quality are not correlated — and are sometimes inversely correlated.
The reason this framework matters for hair loss: the treatments with the strongest evidence are also the cheapest and most boring (generic finasteride and minoxidil), while the treatments marketed most aggressively (exosomes, growth-factor injections, “stem cell therapy”) are typically those with the weakest evidence base. A patient navigating the hair-loss space without an evidence framework ends up paying the most for the things that work the least welltrichology.
A practical hierarchy for 2026:
| Tier | Interventions | Evidence quality | Realistic role |
|---|---|---|---|
| Tier 1 | Finasteride 1mg oral; minoxidil 5% topical | RCT meta-analysismella-fincochrane-min | First-line; foundation of medical management |
| Tier 1.5 | Low-dose oral minoxidil (0.25–5 mg) | Growing dermatology evidencesinclairvano | Specialist-prescribed alternative or adjunct |
| Tier 2 | PRP (platelet-rich plasma) | Mixed RCTsgentilemapar; meta-analysis suggests modest benefitprp-meta | Adjunct to Tier 1; not a replacement for surgery |
| Tier 3 | LLLT (laser caps); mesotherapy | Modest evidence base; limited RCTs | Optional adjunct; effect size small |
| Tier 4 | Exosomes, stem cells; OTC “hair vitamins” without deficiency | Marketing-driven; little controlled evidence | Caution; experimental or unsupported |
Each tier below has its own dedicated cluster article — this pillar gives the framework view.
Tier 1: finasteride and topical minoxidil (the foundation)
The two drugs at the top of the evidence pyramid for AGA management.
Finasteride is an oral 1mg-daily medication that blocks the type II 5-alpha-reductase enzyme, reducing the conversion of testosterone to dihydrotestosterone (DHT) — the androgen that drives follicle miniaturisation in genetically susceptible men. The pooled RCT synthesis (Mella et al., JAMA Dermatology) supports stabilisation of progression in roughly 80–90% of treated men, with measurable density gains in manymella-fin. The drug has been in licensed clinical use for AGA since 1997 (FDA approval) and is on the WHO Model List of Essential Medicines. It is not licensed for use in women of childbearing age (teratogenic risk to a male foetus) but is sometimes used off-label in postmenopausal women with FPHL.
The honest discussion of finasteride side-effects — particularly sexual side-effects, including the controversial post-finasteride syndrome (PFS) — is in the dedicated finasteride article. The summary version: side-effects exist, they’re real, they’re well-documented in the EMA SmPC and FDA Propecia labelema-finfda-fin, they affect a minority of patients, and informed consent is essential.
Topical minoxidil 5% solution is applied twice daily to the scalp. Its mechanism isn’t fully understood — the working hypothesis involves potassium channel opening and prostaglandin pathway modulation — but the empirical evidence is strong: Cochrane RCT support shows modest-to-meaningful density gains in both men and women with AGA over 6–12 months of consistent usecochrane-min. The drug is available without prescription in the UK and has been in use for hair restoration since 1988.
The combination of finasteride plus topical minoxidil is the standard clinical pathway in 2026 for diagnosed male AGA, with each drug producing modest individual effects and the combination producing additive effectsnice-mpb. Both drugs require:
- Consistent daily use — missed doses reduce efficacy substantially
- 4–6 months for visible effect
- 12 months for reasonable assessment
- Indefinite continuation to maintain the effect; AGA progression resumes within 6–12 months of stopping
This isn’t a one-off course of treatment. It’s long-term therapy, like a statin for cholesterol, and the realistic expectation needs to match that frame.
Tier 1.5: low-dose oral minoxidil (the new standard)
Oral minoxidil at low doses (0.25–5 mg daily, depending on patient and indication) has emerged as a standard of care in specialist dermatology centres since the late 2010s. Originally a high-dose antihypertensive medication, low-dose oral minoxidil produces meaningful density gains in AGA at doses well below those associated with cardiovascular side-effects.
The evidence base, growing rapidly:
- Sinclair RD’s initial pilot study (2018) showed effect of low-dose oral minoxidil in female pattern hair losssinclair
- Vañó-Galván S et al. (2021) reported safety data on a cohort of 1,404 patients receiving low-dose oral minoxidil for various hair loss indications, with manageable side-effect profilesvano
- Subsequent dermatology centres in Australia, Spain, and increasingly the UK have adopted oral minoxidil into routine practice
Oral minoxidil’s specific advantages over topical:
- Compliance: a once-daily tablet is easier to take consistently than twice-daily topical application
- No scalp dryness or irritation that some patients experience with topical
- Suitable for patients with topical irritation
- Often combined with finasteride as a complementary mechanism
Side-effect profile at low doses (0.25–5 mg): mild hypertrichosis (increased body hair, mostly at the cheeks and forearms — modest in most patients), occasional ankle swelling, and rare cardiovascular effects that warrant monitoring in the first few monthsvano. The drug is prescription-only in the UK; not available over-the-counter in this formulation.
The full discussion of topical-vs-oral minoxidil is in the dedicated minoxidil article.
Tier 2: PRP (the modest tool)
Platelet-rich plasma (PRP) therapy involves taking the patient’s own blood, centrifuging it to separate the plasma layer rich in platelets and growth factors, and injecting that plasma into the scalp. The proposed mechanism is that the concentrated growth factors stimulate dormant or miniaturising follicles to enter (or stay in) the anagen growth phase.
The evidence is genuinely mixed. The Gentile et al. (2015) RCT showed measurable density gains in the PRP-treated group versus placebogentile. The Mapar et al. (2016) RCT showed no significant effectmapar. The Giordano et al. (2018) systematic review and meta-analysis pooled data across multiple PRP-for-AGA trials and concluded that PRP produces a small-to-moderate increase in hair density relative to controls, with significant heterogeneity in protocol — different PRP preparation methods, volumes, and injection patternsprp-meta.
The honest synthesis: PRP probably has a modest positive effect for some patients, but the effect size is variable, the response is unpredictable, and the typical 3-session-plus-maintenance protocol cost (£700–£1,500 per series) is substantial relative to the benefit. PRP is a useful adjunct to medical management, not a replacement for it. Pairing PRP with continued finasteride and minoxidil is the way it works best in our experience; PRP alone is often disappointing.
What PRP is not: a substitute for hair transplant in advanced AGA. The mechanism only acts on follicles that are still alive but underperforming; it doesn’t restore lost follicles. Patients at moderate-to-advanced stages (Norwood IV+) usually find that PRP plus medical management is insufficient to restore their target density, and the surgical pathway becomes the relevant conversation.
The full discussion of PRP — protocol options, session counts, evidence calibration — is in the dedicated PRP article.
Tier 3: LLLT and mesotherapy (the variable adjuncts)
Low-level laser therapy (LLLT) uses red-light wavelengths (typically 650–680 nm) to stimulate cellular metabolism in scalp follicles. Devices come as in-clinic panels, at-home laser caps (worn for 20–30 minutes per day), and laser combs. The proposed mechanism involves photobiomodulation of follicle stem cells.
The evidence: small RCTs support modest density gains over 6–12 months of consistent usetrichology. Effect size is smaller than finasteride or minoxidil, but the side-effect profile is essentially nil. Useful for patients who can’t or won’t use medical drug therapy, or as a low-friction adjunct. The challenge is compliance: 20–30 minutes daily of cap-wearing is a real time commitment, and many patients drop off after a few months. Devices are also expensive (£300–£800+ for a quality at-home cap).
Mesotherapy for hair loss involves injecting cocktails of vitamins, growth factors, peptides, and other ingredients directly into the scalp at multiple sites. Protocols vary widely between clinics — there’s no standard formulation. Evidence is limited; the ingredients vary so much that meta-analysis is impractical.
Our position: mesotherapy is a vague category. Some specific formulations may have value (particularly when targeting documented deficiencies), but mesotherapy as marketed is often a vehicle for charging high prices for what is essentially intramuscular vitamin delivery. Patients are usually better served by Tier 1 medical management plus targeted nutrient correction (if a deficiency is documented).
Tier 4: exosomes, stem cells, and supplements without deficiency
The bottom of the evidence hierarchy contains the most aggressively-marketed hair-restoration interventions of the 2020s.
Exosomes are small extracellular vesicles released by cells that carry signalling molecules. Exosome therapy for hair loss involves injecting exosomes (typically derived from cultured cells) into the scalp. The marketing claim: exosomes signal follicles to regenerate. The reality, as of 2026: there is no peer-reviewed RCT with appropriate placebo control demonstrating efficacy of exosome therapy for AGA. The interventions are largely promoted via case-series reports, before-and-after photos, and clinic-marketing material. The MHRA in the UK and equivalent regulators internationally have published warnings about unregulated cellular-therapy products being marketed for cosmetic indications without supporting evidence. Until appropriate trials are published, our position is that exosomes for hair loss are largely unproven and patients considering them should understand they’re paying for an experimental intervention.
“Stem cell” hair therapy is similarly variable. The phrase is used loosely — some clinics inject patient-derived adipose-tissue products, others inject commercial preparations of unclear cellular content, others use the language metaphorically. Genuine stem-cell therapy with proper cellular characterisation, regulatory approval, and RCT support does not currently exist for AGA in the consumer cosmetic market.
OTC “hair vitamins” and supplements are a separate category. The honest position:
- If you have a documented deficiency (low ferritin, low vitamin D, hypothyroidism, low B12), correcting that deficiency typically resolves any deficiency-related hair sheddingnhs
- If you don’t have a documented deficiency, generic “hair vitamins” sold over-the-counter are unlikely to produce meaningful results
- The market is large and the marketing claims are far ahead of the evidence
Specific evidence-supported nutrient interventions (e.g., iron supplementation in iron-deficient women with hair shedding) are useful. Generic “boost your hair growth” supplements without a deficiency present are unlikely to be useful.
How to combine treatments (clinical protocols)
The realistic 2026 clinical protocol for diagnosed AGA:
Stage 1: Newly diagnosed AGA, Norwood II–III
- Finasteride 1mg oral daily (subject to side-effect discussion and informed consent)
- Topical minoxidil 5% twice daily OR low-dose oral minoxidil if specialist-prescribed
- Photographic monitoring every 6 months in identical conditions
- Review at 12 months for response assessment
- Optional: PRP series (3 sessions over 3 months, then 6-monthly maintenance) as adjunct
Stage 2: Moderate AGA, Norwood IV–V, or non-responders to Stage 1
- Continue finasteride and minoxidil
- Add or continue PRP as adjunct
- Hair transplant assessment — surgery becomes a sensible parallel conversation (full pathway in the hair transplant pillar)
- Optional: LLLT as compliance-friendly adjunct
Stage 3: Advanced AGA, Norwood VI–VII
- Hair transplant is the primary tool at this stage; medical management alone is insufficient to restore target density
- Continue finasteride and minoxidil to protect remaining native hair
- PRP and LLLT roles are minimal at this stage
The order matters: medical management is the foundation regardless of stage because it acts on the underlying disease process. Procedures (PRP, LLLT) are adjuncts. Surgery is the tool for restoring lost density at moderate-to-advanced stages, but it doesn’t change AGA — which is why finasteride and minoxidil typically continue indefinitely after surgery as well.
When non-surgical isn’t enough
Non-surgical management has natural limits. For most patients, the conversation about transitioning to (or adding) hair transplant happens when:
- Norwood stage has reached IV+ despite consistent medical management for 12+ months
- Visible density has fallen below the patient’s target despite Tier 1 therapy
- Donor density is sufficient to make surgical intervention worthwhile (covered in the AGA article)
- The patient’s preferences prioritise restored visible density at the cost of a one-time surgical intervention
Surgery isn’t a replacement for medical management — most surgeons recommend continuing finasteride and minoxidil after surgery to protect the result. The transplanted follicles are largely permanent; the surrounding native follicles still need ongoing protection from AGA progression.
For UK patients, the BergemHealth two-clinic model offers parity-priced surgical pathways at JCI-accredited Liv Hospital Ulus in Istanbul and CQC-registered 99 Harley Street in London — same per-graft pricing, two recovery options. (Detail in the choosing-a-clinic pillar and Istanbul vs London logistics article.)
Side-effect transparency
A short note on the side-effect approach across this pillar and its cluster articles. We treat side-effects as essential clinical information to share, not as marketing inconvenience to minimise:
- Finasteride: real sexual side-effects in a minority of patients, full EMA SmPC and FDA Propecia label data citedema-finfda-fin; PFS controversy discussed without dismissalpfs; informed consent is essential
- Minoxidil: hypertrichosis (extra body hair), mild scalp irritation (topical), ankle swelling and rare cardiovascular effects (oral); shedding phase in first 8–12 weeks of use is normal and not a treatment failure
- PRP: minor injection site discomfort, transient redness, very rare infection risk; main “side-effect” is the cost and the modest effect size relative to expectation
- Surgery: covered in detail in recovery and aftercare and surgeon safety
A patient making an informed decision needs the side-effect picture honestly. Clinics that minimise side-effects to close the sale are doing the patient a disservice.
How BergemHealth approaches non-surgical treatment
BergemHealth’s first-consultation pathway includes a discussion of non-surgical options at every appointment, regardless of whether the patient came in for a transplant assessment. Most early-stage patients are better served by medical management first, and many moderate-stage patients are best served by combined medical-plus-surgical pathways rather than surgery alone.
The Harley Street consulting team — led by Dr. Sumeyye Yuksel, GMC-registeredgmc — handles consultations, prescriptions, and follow-up reviews for non-surgical pathways. Patients can be seen for medical-management-only consultations without any surgical commitment. Pricing for non-surgical treatment is at standard UK private dermatology rates; medication scripts can be filled at any UK pharmacy.
PRP procedures, when appropriate as an adjunct, are performed at both Harley Street (London) and Liv Hospital Ulus (Istanbul) by clinical staff under supervision. The protocol is standardised across the network so that a patient can begin a PRP series in one location and continue in the other.
For surgical pathways when they become appropriate, Dr. Hamid Aydın leads at Liv Hospital Ulus (ISHRS member, 25,000+ procedures since 2000)ishrs, with the same pricing on both sides of the network: from £1,250 (Standard FUE), £1,750 (Sapphire FUE), £2,250 (Direct DHI).
What to do next
If you’re at early-stage AGA and want to start medical management, see the dedicated articles: finasteride efficacy and side-effects and topical vs oral minoxidil. For PRP details, see the PRP article.
For diagnosis if you’re not sure what kind of hair loss you have, the hair loss pillar has the diagnostic decision tree. For androgenetic alopecia specifically, the lookalikes and how to tell them apart.
If you’ve worked through medical management and are considering surgery, request a free assessment from BergemHealth’s London or Istanbul team. The consultation includes a discussion of where you are in the medical-management pathway, whether non-surgical options have been adequately tried, and whether the surgical pathway is the right next step. CQC-registered facility in London. JCI-accredited hospital in Istanbul. ISHRS-member lead surgeon.
Frequently asked questions
What is the best non-surgical hair loss treatment?
For most men with diagnosed AGA in 2026, the combination of finasteride 1mg daily plus topical minoxidil 5% twice daily is the foundation, with RCT-level support for both drugsmella-fincochrane-min. For women with FPHL, topical minoxidil is first-line; finasteride is contraindicated in women of childbearing age. Low-dose oral minoxidil is increasingly used in specialist centres as an alternative or complement.
Does PRP work for hair loss?
PRP probably has a modest positive effect for some patients, but the RCT evidence is genuinely mixed (Gentile 2015 positivegentile, Mapar 2016 nullmapar; Giordano 2018 meta-analysis reports a small-to-moderate pooled benefitprp-meta) and the response is unpredictable. PRP works as an adjunct to medical management, not a replacement for it, and doesn’t substitute for surgery in advanced AGA. Pair PRP with continued finasteride and minoxidil for best chance of meaningful effect.
Should I take finasteride or minoxidil?
Both, in most cases. They have different mechanisms (finasteride blocks DHT; minoxidil extends anagen) and produce additive effects. Topical minoxidil is OTC; finasteride is prescription-only and requires a side-effect discussion. Specific patient factors (sexual side-effect concerns with finasteride; topical irritation tolerance with minoxidil) inform the individual decision.
Do exosomes work for hair loss?
Not at a level supported by RCT evidence. As of 2026, there is no peer-reviewed RCT with appropriate placebo control demonstrating efficacy of exosome therapy for AGA. The interventions are promoted via case-series reports and clinic marketing rather than controlled trials. Until appropriate trials are published, exosomes for hair loss are largely unproven and patients should understand they’re paying for an experimental intervention.
Is mesotherapy effective for hair?
Mesotherapy as currently marketed is a vague category — protocols vary widely between clinics and there’s no standard formulation. The evidence base for routine mesotherapy as AGA management is weak. Specific evidence-supported nutrient correction (e.g., iron supplementation when iron-deficient) is useful; mesotherapy injecting generic “growth factor cocktails” without a defined indication has limited supporting evidence.
Can hair loss be treated without transplant?
Yes, at early to moderate stages. Most adult AGA can be slowed or partially reversed with finasteride and minoxidil started before substantial miniaturisation has occurred. PRP and LLLT can be added as adjuncts. At moderate-to-advanced stages (Norwood IV+), medical management alone is usually insufficient to restore target density, and surgery becomes a sensible parallel conversation.
What’s the order of hair loss treatments?
Diagnose first (rule out telogen effluvium, alopecia areata, scarring alopecias, deficiencies). For confirmed AGA: start with finasteride + topical or oral minoxidil. Add PRP as adjunct if desired. Add LLLT for compliance-friendly home maintenance. Reassess at 12 months. Consider surgery at moderate-to-advanced stages while continuing medical management.
Will hair vitamins help me grow more hair?
If you have a documented deficiency (low ferritin, vitamin D, B12, hypothyroidism), correcting it typically resolves deficiency-related shedding. If you don’t have a deficiency, generic OTC hair vitamins are unlikely to produce meaningful results. The marketing of generic supplements is far ahead of the evidence base. Blood work is the way to find out if a specific deficiency applies to you.
How long do I need to take finasteride or minoxidil?
Indefinitely, to maintain the effect. AGA is progressive; both drugs slow the process while taken and the process resumes within 6–12 months of stopping. This isn’t different from how a statin works for cholesterol — it’s long-term therapy. Stopping after a year typically loses much of the gain.
Are there hair loss treatments specifically for women?
Topical minoxidil 5% (or 2% formulations sometimes used to reduce hypertrichosis risk) is first-line for FPHL. Low-dose oral minoxidil is increasingly used by specialists. Spironolactone (an anti-androgen) is sometimes prescribed. Finasteride is generally contraindicated in women of childbearing age but is used off-label in postmenopausal women in some specialist centres. PRP, LLLT, and treatment of underlying medical conditions (PCOS, thyroid disease) are all relevant. The picture is more individualised in women than in mennice-flu.
Can I combine surgical and non-surgical treatment?
Yes, and this is the recommended approach for most moderate-to-advanced AGA. Surgery restores density at affected zones; medical management protects the surrounding native hair from continued miniaturisation. Stopping finasteride and minoxidil after surgery is associated with progression of native AGA in subsequent years, which can leave the surgical result looking isolated as the surrounding hair recedes.
Sources
- NHS — Hair loss overview. https://www.nhs.uk/conditions/hair-loss/ ↩
- NICE Clinical Knowledge Summary — Male pattern baldness. https://cks.nice.org.uk/topics/male-pattern-baldness/ ↩
- NICE Clinical Knowledge Summary — Female pattern hair loss. https://cks.nice.org.uk/topics/female-pattern-hair-loss/ ↩
- Institute of Trichologists (UK) — Position statements. https://www.trichologists.org.uk/ ↩
- Mella JM, Perret MC, Manzotti M, et al. “Efficacy and safety of finasteride therapy for androgenetic alopecia: a systematic review.” Arch Dermatol. 2010;146(10):1141-1150. DOI: 10.1001/archdermatol.2010.256 ↩
- Gupta AK, Charrette A. “Topical minoxidil for androgenetic alopecia.” Cochrane Database Syst Rev. DOI: 10.1002/14651858.CD007628 ↩
- Sinclair RD. “Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil.” Int J Dermatol. 2018. DOI: 10.1111/ijd.13838 ↩
- Vañó-Galván S et al. “Safety of low-dose oral minoxidil for hair loss: a multicentre study of 1,404 patients.” J Am Acad Dermatol. 2021. DOI: 10.1016/j.jaad.2020.06.1023 ↩
- Gentile P et al. “PRP for androgenetic alopecia: randomised controlled trial.” Stem Cells Transl Med. 2015. DOI: 10.5966/sctm.2015-0107 ↩
- Mapar MA et al. “Efficacy of platelet-rich plasma for hair regrowth: a placebo-controlled study.” Iran J Dermatol. 2016. ↩
- Giordano S, Romeo M, Lankinen P. “Platelet-rich plasma for androgenetic alopecia: a systematic review and meta-analysis.” Plast Reconstr Surg Glob Open / Dermatol Surg. 2018. https://pubmed.ncbi.nlm.nih.gov/29649029/ ↩
- European Medicines Agency — Finasteride 1 mg Summary of Product Characteristics. https://www.ema.europa.eu/ ↩
- US Food and Drug Administration — PROPECIA (finasteride) 1 mg tablets prescribing information (2022). https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/020788s030lbl.pdf ↩
- Trüeb RM. “Discussion on post-finasteride syndrome.” Skin Appendage Disord. 2018. DOI: 10.1159/000490031 ↩
- International Society of Hair Restoration Surgery — Member directory. https://www.ishrs.org/ ↩
- General Medical Council — The Medical Register. https://www.gmc-uk.org/registration-and-licensing/the-medical-register ↩
Как BergemHealth подходит к этому
Операции выполняются в Liv Hospital Ulus (Стамбул) с аккредитацией JCI доктором Hamid Aydın и резидентной хирургической командой. Консультация в Великобритании и 12-месячный уход в нашем офисе на Harley Street с регулированием CQC. Прозрачные цены и бесплатная корректировка при необходимости.
Бесплатная консультация →