Finasteride for Hair Loss: Efficacy, Side Effects, Honest Assessment
Finasteride explained: how it works, RCT efficacy, sexual side-effects, post-finasteride syndrome (PFS), and who should and shouldn't consider it.
Medical disclaimer. This article is educational and not medical advice. Finasteride is a prescription medication. Decisions about starting, continuing, or stopping it should be made with a clinician who has reviewed your individual medical history.
Quick answer
Finasteride is an oral 1mg-daily medication that blocks the 5-alpha-reductase enzyme, reducing the conversion of testosterone to dihydrotestosterone (DHT) — the androgen driving follicle miniaturisation in androgenetic alopecia. Efficacy: stabilises AGA progression in roughly 80–90% of treated men, with measurable density gains in many over 12 months of consistent use, supported by RCT meta-analysis (Mella et al., JAMA Dermatology). Side effects: sexual side-effects (decreased libido, erectile dysfunction, ejaculatory dysfunction) occur in roughly 1–4% of patients in pivotal RCT data, with EMA and FDA labelling now including post-marketing reports of persistent symptoms in some patients (post-finasteride syndrome, PFS). This article walks the efficacy data, the full side-effect picture, and who should and shouldn’t consider it.
Table of contents
- What finasteride is and how it works
- The efficacy evidence
- The side-effect profile (the honest version)
- Post-finasteride syndrome (PFS) — without dismissal
- Who should consider finasteride
- Who should not
- How to start, monitor, and stop
- Finasteride vs dutasteride
- How BergemHealth approaches finasteride prescribing
- What to do next
- Frequently asked questions
What finasteride is and how it works
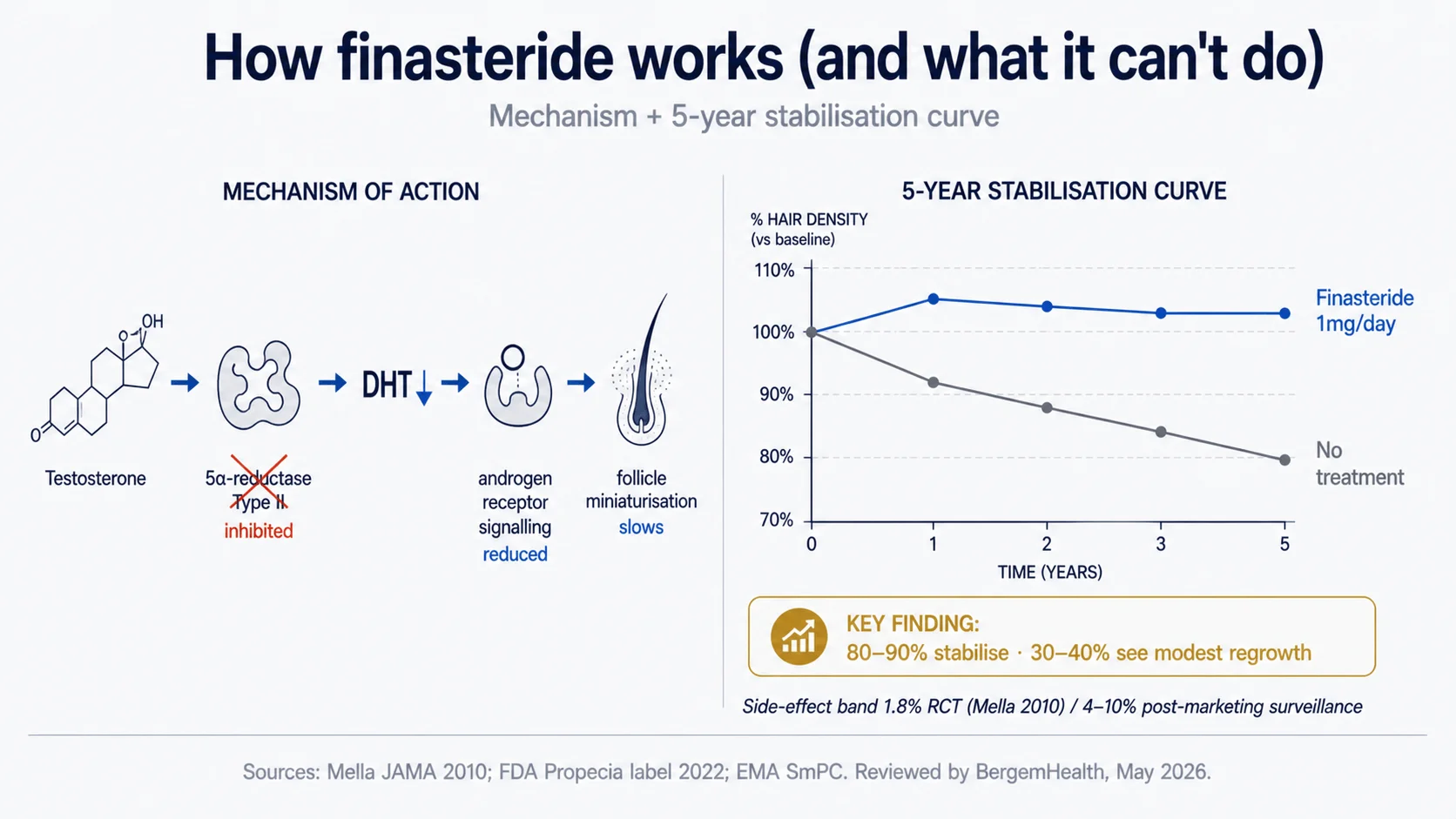
Definition. Finasteride is a synthetic 4-azasteroid that selectively inhibits the type II 5-alpha-reductase enzyme. By blocking this enzyme, finasteride reduces the conversion of testosterone to dihydrotestosterone (DHT) — the more potent androgen responsible for follicle miniaturisation in androgenetic alopecia. The 1mg daily dose used for hair loss reduces serum DHT by roughly 65–70% while leaving testosterone levels essentially unchanged.
A few facts to anchor:
- First approved: 1992 by the FDA at 5mg dose for benign prostatic hyperplasia; 1997 at 1mg dose for AGA in menfda-fin
- Brand names: Propecia (1mg, AGA); Proscar (5mg, BPH); generic finasteride 1mg widely available in the UK and globally since 2014
- UK prescription: Schedule POM (prescription-only); typically prescribed by GP or private dermatology clinic
- Mechanism specificity: type II 5-alpha-reductase inhibitor; doesn’t block the type I isoform (which dutasteride does)
- Cost in UK: generic 1mg approximately £10–£20 per month with NHS prescription or as a private script
Finasteride doesn’t change the underlying genetic susceptibility to AGA — it modifies the hormonal driver of miniaturisation, slowing or partially reversing the process while taken. The drug effect persists as long as it’s taken; AGA progression resumes within 6–12 months of stoppingmella-fin.
The efficacy evidence
The pooled RCT synthesis on finasteride for male AGA (Mella et al., systematic review of placebo-controlled trials)mella-fin:
- Stabilisation of progression in roughly 80–90% of men
- Measurable density gains at 12 months in roughly 65% of treated men
- Effect size in pivotal trials: average density increase of approximately 7% at vertex, with smaller gains at frontal scalp at 12 months
- 5-year follow-up data showing sustained effect with continued use
- Most pronounced effect at vertex (crown); modest at frontal hairline
- Effect plateau around 12–24 months of treatment
The 80–90% stabilisation rate is the most commonly cited figure and well-supported across the literature. The “measurable density gain” component is more variable — some patients show substantial regrowth, others stabilise without significant gain. Earlier age of starting and earlier Norwood stage at start both correlate with better outcomes.
In women: finasteride is not licensed for treatment of female pattern hair loss in most jurisdictions, primarily because of teratogenic risk to a male foetus. Off-label use in postmenopausal women is reported in specialist literature with mixed efficacy. The mainstream non-surgical approach for FPHL is topical or low-dose oral minoxidil (covered in the minoxidil article).
Time course of effect: 3–6 months for stabilisation of shedding to become apparent; 6–12 months for density gains to be visible; 12 months as standard assessment point. Slow responders sometimes show effect at 18–24 months. Patients who don’t tolerate the drug typically know within the first 3 months.
The side-effect profile (the honest version)
This section follows the EMA Summary of Product Characteristics (SmPC) for finasteride 1mg and the FDA Propecia prescribing labelema-finfda-fin, plus post-marketing surveillance literature. Neither minimised nor exaggerated.
Sexual side-effects — the most-discussed category:
- Decreased libido: 1.8% of treated men vs 1.3% placebo in pivotal RCT data (FDA label); 4–10% in some post-marketing surveillance studies
- Erectile dysfunction: 1.3% of treated vs 0.7% placebo in pivotal trials; higher rates in some surveillance data
- Ejaculatory disorders (decreased volume, decreased sensation): 0.8% of treated vs 0.4% placebo in pivotal trials
- In most patients, sexual side-effects resolve within weeks of stopping the drug
The pivotal RCT figures are the conservative end; post-marketing surveillance figures are higher and reflect real-world reporting bias and a broader patient population. The honest reading: sexual side-effects affect a meaningful minority, with conservative estimates around 2–4% experiencing some sexual symptom and a smaller proportion reporting symptoms that materially affect quality of life.
Other side-effects in EMA SmPC and FDA label:
- Mood changes including depression: reported in post-marketing data; incidence not well-quantified
- Breast tenderness or enlargement: rare
- Reduction in serum PSA: relevant for men over 50 (finasteride approximately halves PSA, which screening labs need to know)
- Allergic reactions, hypersensitivity, angio-oedema: rare to very rare
Pregnancy and women: finasteride is teratogenic to a male foetus. Women who are pregnant or may become pregnant should not handle crushed or broken finasteride tablets. Standard 1mg coated tablets are safe to handle while intact.
This data is what informed consent requires before prescribing — and what some clinic marketing minimises to the point of misrepresentation.
Post-finasteride syndrome (PFS) — without dismissal
Post-finasteride syndrome (PFS) refers to a cluster of persistent symptoms — sexual dysfunction, depression, cognitive changes — reported by some men after stopping finasteride. Defining feature: symptoms that fail to resolve in the expected timeframe (within weeks of stopping) and may persist for months or years.
The clinical and regulatory picture as of 2026:
- EMA and FDA labelling has been updated multiple times to include post-marketing reports of persistent symptoms in some patientsema-finfda-fin
- Trüeb RM and others have published clinical discussions characterising PFS as a real but poorly-understood syndromepfs
- Mechanistic hypotheses include neurosteroid pathway disruption and persistent androgen-receptor sensitisation — none definitively confirmed
- Estimated prevalence is genuinely unknown; estimates range widely depending on study population and definition
Our position: PFS is a phenomenon with enough clinical and regulatory recognition that informed consent should mention it as a possibility. Dismissing it entirely is inconsistent with the EMA and FDA having included persistence reports in the official label. Conversely, presenting it as a high-probability outcome of starting finasteride would be an over-reading of the data.
The practical approach: know it’s a possibility referenced in EMA and FDA labelling; stop the drug at the first sign of symptoms that don’t quickly resolve (earlier intervention may reduce the chance of persistence); seek specialist input rather than re-trying or doubling down if symptoms persist after stopping; consider topical or low-dose oral minoxidil as alternative if finasteride risk feels too high (covered in the minoxidil article).
Who should consider finasteride
Strong candidates:
- Diagnosed androgenetic alopecia in men at Norwood II–V where stabilising or reversing progression is the goal
- Patients comfortable with long-term medication — finasteride needs to be taken indefinitely to maintain effect
- Patients without significant baseline depression or sexual dysfunction — these can be exacerbated and complicate side-effect attribution
- Younger men with progressive AGA who’ll benefit from many years of stabilisation
- Patients post-hair-transplant who want to protect native hair density alongside the surgical result
The risk-benefit is most favourable when the AGA is genuinely progressing (the patient has something to lose without treatment) and the patient is in good health without complicating factors.
Who should not
Less suitable or contraindicated:
- Women who are pregnant, may become pregnant, or breastfeeding — teratogenic risk
- Premenopausal women with FPHL — finasteride is not licensed for this indication; topical minoxidil is first-line
- Patients with significant baseline depression — case-by-case assessment with a psychiatrist or GP recommended
- Patients with significant baseline sexual dysfunction — case-by-case
- Patients who experienced significant side-effects on prior finasteride exposure — re-trial generally not advised
- Patients planning male fertility in the next 6–12 months — finasteride can affect semen parameters in a minority of men
- Patients undergoing PSA monitoring for prostate cancer screening — possible but the prescribing GP and the screening lab need to know about the finasteride
How to start, monitor, and stop
Starting: clinical consultation with discussion of side-effect profile and written informed consent; prescription 1mg daily oral, no food restrictions; check-in at 3 months for any side-effects; 6-month and 12-month reviews to assess efficacy with photographs.
Monitoring during treatment: self-monitoring for sexual or mood side-effects (particularly first 6 months); PSA testing if over 50 with the lab informed about finasteride use (multiplier of 2× applied); no routine blood-work monitoring required for standard 1mg use.
Stopping: stop immediately if sexual or mood symptoms develop and don’t resolve within a few weeks; planned discontinuation generally fine without taper (no withdrawal pharmacology); AGA progression typically resumes within 6–12 months of stopping; visible loss of gained density usually within 12–18 months.
The discontinuation question is itself a useful framing: would you be willing to take this drug long-term? Patients who say “no, I’d want to stop in 12 months” should consider whether the pathway makes sense — the gains made will be lost when the drug stops.
Finasteride vs dutasteride
Dutasteride inhibits both type I and type II 5-alpha-reductase (versus finasteride’s type-II-only): stronger DHT suppression (~93% vs ~70%), slightly stronger efficacy in clinical trials, longer half-life (~5 weeks vs ~6 hours), similar side-effect profile with some evidence of slightly higher sexual side-effect rates. In the UK, dutasteride is licensed for benign prostatic hyperplasia, not for AGA — off-label prescribing for AGA happens in specialist dermatology settings. The longer half-life means symptoms following discontinuation can persist longer than with finasteride. For most patients, finasteride is the appropriate first-line choice; dutasteride is sometimes used in finasteride non-responders.
How BergemHealth approaches finasteride prescribing
The GMC-registered consulting team at 99 Harley Street, London prescribes finasteride for diagnosed male AGA after a structured consultation: clinical history (AGA confirmation, family history, current medications, baseline mood and sexual function); trichoscopy to rule out lookalikes; side-effect discussion covering the EMA SmPC, post-marketing surveillance, and PFS literature; written informed consent; 3-, 6-, and 12-month reviews in person or remotely.
Patients who choose not to start finasteride after the consultation are common — and that’s a valid outcome. Topical or low-dose oral minoxidil is available as alternative or starting point. PRP, LLLT, and ongoing photographic monitoring are options for active management without finasteride.
For patients on the surgical pathway, continuing finasteride post-transplant to protect native hair is the standard recommendation. Dr. Hamid Aydın’s surgical team at Liv Hospital Ulus, Istanbulishrs coordinates with the medical management team for consistency of approach across the BergemHealth two-clinic network. Dr. Sumeyye Yuksel leads the GMC-registered consulting team at Harley Streetgmc.
What to do next
For the broader non-surgical framework, see the hair-treatment pillar. For the alternative or complementary medication, see the minoxidil article. For PRP as an adjunct procedure.
For diagnosis if you’re not yet sure what kind of hair loss you have, the hair loss pillar. For androgenetic alopecia specifically — the condition finasteride is treating. For staging via the Norwood scale.
If you’re considering finasteride and want a personalised assessment with proper informed consent, request a free assessment from BergemHealth’s London team. The consultation includes the clinical history, trichoscopy, side-effect discussion, and written prescription pathway under GMC-registered supervision. CQC-registered facility in London. JCI-accredited hospital in Istanbul. ISHRS-member lead surgeon.
Frequently asked questions
Does finasteride work for hair loss?
Yes, with strong evidence. RCT meta-analysis (Mella et al.) supports stabilisation of AGA progression in roughly 80–90% of treated men, with measurable density gains in a majority over 12 monthsmella-fin. Effect is most pronounced at vertex (crown), modest at frontal hairline. Most patients see stabilisation by 6 months and density gains by 12 months.
What are finasteride side effects?
Sexual side-effects (decreased libido, erectile dysfunction, ejaculatory disorders) are reported in roughly 1–4% of treated men in pivotal RCT data, higher in some post-marketing surveillanceema-finfda-fin. Other reported side-effects include mood changes, breast tenderness, and rare hypersensitivity reactions. EMA and FDA labelling includes post-marketing reports of persistent symptoms in some patients (PFS).
How long does finasteride take to work?
Stabilisation of shedding becomes apparent at 3–6 months. Density gains begin to be visible at 6–12 months. Standard assessment point is 12 months. Slow responders sometimes show effect at 18–24 months. The full effect plateau is around 12–24 months of treatmentmella-fin.
Can finasteride cause permanent side effects?
EMA and FDA labelling includes post-marketing reports of persistent sexual and mood symptoms in some patients (post-finasteride syndrome). Estimated prevalence is genuinely unknown. The precautionary approach: stop the drug at first sign of symptoms that don’t quickly resolve; seek specialist input if symptoms persist after stoppingpfs.
Should I take finasteride at 25?
This is an individual decision. The case for early start: AGA is progressive, finasteride works better when started before substantial miniaturisation, starting at 25 gives many years of stabilisation. The case for waiting: side-effect risk is real, visible benefit at Norwood II is small, and starting later remains an option. The decision should be made with a clinician who has reviewed your individual case.
What is post-finasteride syndrome?
A cluster of persistent symptoms — sexual dysfunction, depression, cognitive changes — reported by some men after stopping finasteride, with symptoms that fail to resolve in the expected timeframe. EMA and FDA labelling has been updated to include post-marketing reports of persistence; mechanism is poorly understood. The phenomenon has enough clinical and regulatory recognition to warrant mention in informed consentpfs.
Can I stop finasteride?
Yes — generally fine to stop without taper (no withdrawal pharmacology). The consequence: AGA progression typically resumes within 6–12 months; gained density is usually lost within 12–18 months. If you stop because of side-effects, most resolve within a few weeks to months — though EMA and FDA labelling includes post-marketing reports of persistent symptoms in some patients.
Finasteride vs dutasteride — which is better?
Dutasteride suppresses DHT more strongly (~93% vs ~70%) and shows marginally stronger efficacy. Finasteride is licensed for AGA in the UK; dutasteride is licensed only for BPH and is used off-label for AGA in specialist settings. For most patients finasteride is the appropriate first-line choice due to licensing and stronger evidence base; dutasteride is sometimes used in non-responders or specialist contexts.
Can finasteride cause depression?
Mood changes including depression are reported in post-marketing data, with incidence not well-quantifiedema-finfda-fin. Patients with significant baseline depression should discuss with their clinician before starting. Patients on finasteride who develop new mood symptoms should consider whether stopping the drug resolves them.
Can I take finasteride with minoxidil?
Yes — and this is the standard combination pathway. Different mechanisms produce additive effects. The combination is well-tolerated and is the foundation of medical management for diagnosed male AGA in 2026. Minoxidil can be topical or low-dose oral; both combine with finasteride safely.
Sources
- Mella JM, Perret MC, Manzotti M, et al. “Efficacy and safety of finasteride therapy for androgenetic alopecia: a systematic review.” Arch Dermatol. 2010;146(10):1141-1150. DOI: 10.1001/archdermatol.2010.256 ↩
- European Medicines Agency — Finasteride 1 mg Summary of Product Characteristics. https://www.ema.europa.eu/ ↩
- US Food and Drug Administration — PROPECIA (finasteride) 1 mg tablets prescribing information (2022). https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/020788s030lbl.pdf ↩
- Trüeb RM. “Discussion on post-finasteride syndrome.” Skin Appendage Disord. 2018. DOI: 10.1159/000490031 ↩
- NICE Clinical Knowledge Summary — Male pattern baldness. https://cks.nice.org.uk/topics/male-pattern-baldness/ ↩
- International Society of Hair Restoration Surgery — Member directory. https://www.ishrs.org/ ↩
- General Medical Council — The Medical Register. https://www.gmc-uk.org/registration-and-licensing/the-medical-register ↩
Як BergemHealth підходить до цього
Операції виконуються у Liv Hospital Ulus (Стамбул) з акредитацією JCI д-ром Hamid Aydın та резидентною хірургічною командою. Консультації у Великій Британії та 12-місячний догляд у нашому офісі на Harley Street з регулюванням CQC. Прозорі ціни та безкоштовна корекція за потреби.
Безкоштовна консультація →